|
Getting your Trinity Audio player ready... |
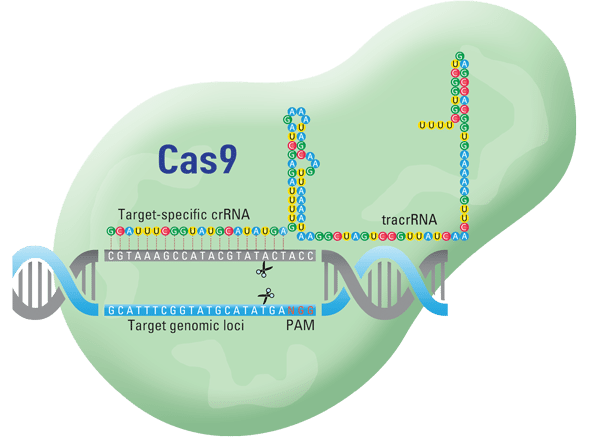
Download this post... Tiny vials of recently repaired blood cells are thriving in a Stanford incubator, proof that a powerful new gene-editing technique is fixing errant genes that cause so much human suffering. Until recently, gene therapy was laborious, crude and unsafe for human testing. But the new technology, called CRISPR-Cas9, acts as a microscopic...